Resolution Biomedical Obtains ISO 13485 Certification
Internationally-recognized standard opens the door to registration in multiple countries.
TUSTIN, CALIFORNIA – Resolution Biomedical, Inc. announced today that it has received ISO 13485 Certification for “design and development, manufacture, distribution of liquid-based cytology test kits for use in the clinical and laboratory setting.”
ISO 13485 follows standards promulgated by the International Organization for Standardization (ISO) for the design and manufacture of medical devices. Certification was issued following an audit of the Resolution Biomedical’s manufacturing processes and quality management system.
“ISO 13485 certification is an important milestone for us, as it validates our commitment to quality manufacturing, and allows us to pursue registration in many international markets,” announced Michael Friedl, CEO of Resolution Biomedical.
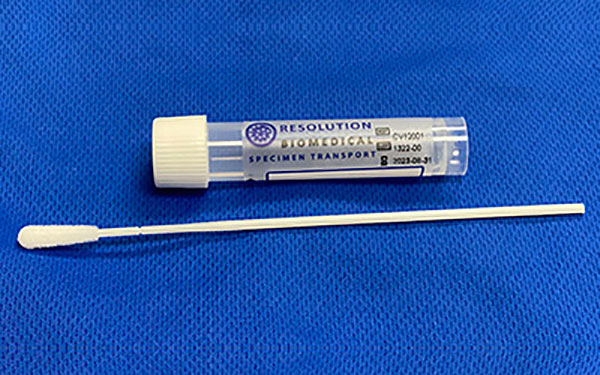
In addition to being registered with the US FDA & CE Marked, ClearPrep Next-Generation Liquid-Based Cytology has been validated for all types of cytology, which is used to test for certain types of cancer, including cervical, oral, bladder, thyroid, among others. ClearPrep has also been validated on leading molecular-based HPV/STI testing platforms. ClearPrep features a unique patentpending collection vial that improves patient outcomes by increasing cellularity of samples at the
point of collection.
Please see www.clearprepcytology.com for further information and distribution contacts.
About Resolution Biomedical, Inc.
Resolution Biomedical was founded in 2009 to develop and launch ClearPrep Next-Generation Liquid-Based Cytology. ClearPrep is used by medical testing laboratories for preparation of samples for screening of several types of cancer. It is currently available in the United States and multiple countries around the world. The company is licensed by the California Department of Public Health, is ISO 13485 certified and its facility has been registered with the US FDA.
Contact: Terence Lynch